Heat always flows from substances of a warmer temperature to substances of a cooler temperature. Heat (thermal energy) will be exchanged in an environment until a common temperature (equilibrium) is achieved. This equalizing of temperature in an environment is caused by a combination of three basic types of heat transfer: conduction, radiation and convection.
Conduction
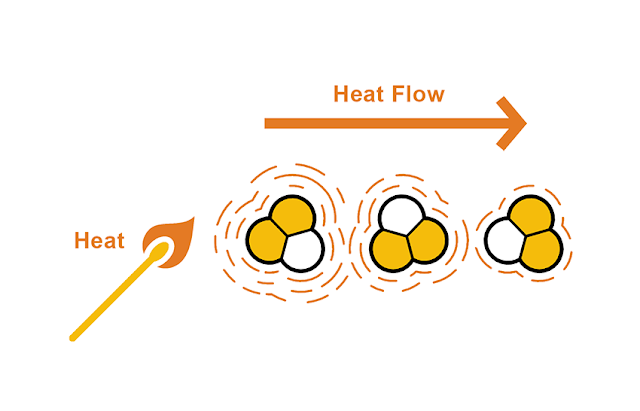
Conduction is the transfer of vibrating energy between adjacent molecule (Figure 1). The transfer is always from the warmer region (i.e., faster vibration) to the cooler region (i.e., slower vibration). Conductive heat transfer can happen in any direction and is independent of gravity.
If a person place the palm of a hand flatly upon a surface, that surface will feel relatively warmer or cooler. Such a sensation is indicative of conductive heat transfer. A familiar demonstration of conduction is holding a mug filled with hot coffee. The hot coffee causes the molecules of the mug to vibrate more rapidly. Heat, therefore, is transmitted and soon thereafter enters the hand holding the mug. The faster (warmer) molecules will continue to transfer heat to the mug and hand until equilibrium is achieved.
How readily a substance conducts heat depends on its molecular structure. Typically, denser materials (e.g., concrete or steel) will conduct heat more readily than less dense materials (e.g., wood or mineral wool). Keep in mind that conduction depends on the transfer of vibrating energy between adjacent molecules. No molecules, no conduction. This is precisely why a near-vacuum space (such as the air space in an insulated glass unit) is effective at reducing heat transfer.
|
Figure 1: Heat transfer by conduction. Illustration courtesy of Daniel Overbey. Adapted from Moore, 1993. |
Radiation
Radiation is the transfer of molecular vibrating energy (heat) by electromagnetic waves (Figure 2). As the molecules along the surface of a substance vibrate, electromagnetic waves (radiation) travel away from the surface at the speed of light. These waves travel until they strike a surface and are absorbed. The molecules on the receiving surface absorb some of the radiant energy and converts it into heat.
All substances radiate energy in all directions as a function of the absolute surface temperature. This means that even a cool surface will give off radiation at the same time that it is absorbing radiation. The net radiant energy transfer is from the warmer surface to the cooler surface.
|
Figure 2: Radiant energy transfer between two surface molecules. Illustration courtesy of Daniel Overbey. Adapted from Moore, 1993. |
Convection
As a substance is heated, its molecules vibrate increasingly faster. Generally, in response to this increase in activity, the molecules separate themselves more and more. The additional space between the molecules results in an expansion of the substance. This in turn causes solids and liquids to increase in volume. It also causes fluids (liquids and gases) become less dense and, therefore, more buoyant than adjacent cooler fluids. Convection is the transfer of heat via a moving fluid medium (Figure 3).
Technically, convection is a form of both radiation and conduction in combination with the movement of the fluid as more dense (cooler) fluid is weighed down by gravity; but for purposes of understanding heat transfer as related to the built environment, it is useful to consider convection as a separate mode of heat transfer.
Side note: The phenomenon of warmer fluids being more buoyant explains how a hot air balloon works. However, it has also "given rise" (sorry) to the oversimplified assertion that heat rises. Technically, heat does not rise.
|
Figure 3: Heat transfer from location "A" to location "B" by convection. Illustration courtesy of Daniel Overbey. Adapted from Moore, 1993. |
But wait, there's more!
These is actually a fourth form of heat transfer: phase change. It takes energy to convert a solid into a liquid or a liquid into a gas. However, the energy inputs required for phase change to occur are not able to be perceived as a change in sensible heat (i.e., increasing/decreasing molecular vibration). Rather, this latent heat is embodied by the phase change itself.
Reference:
Moore, Fuller, 1993, Environmental Control Systems: Heating Cooling Lighting. McGraw-Hill, Inc., pp. 13-22.




Recent Comments
These are actually very helpful tips. It is...
This is the most beneficial blog for all...
This blog is a great resource for anyone...
Thank you for sharing this important information. I...
This is a very interesting subject of the...